European cardiology is poised for a potential leap forward as clinical studies progress on an innovative cardiac catheter designed to treat arrhythmia with unprecedented speed and precision. Developed and manufactured within the European Union, the device represents a significant Made in Europe advancement in medical technology aimed at tackling a condition affecting millions across the continent.
Atrial fibrillation and other heart rhythm disorders impact approximately one in ten Europeans aged 60 and over. The symptoms range from unnoticeable to debilitating. "Clinical perception of the condition is highly variable," explains Petr Neužil, Head of the Cardiology Clinic at Prague's Na Homolce Hospital. "The patient may perceive nothing; they may be completely asymptomatic. Then, they can feel palpitations. They can feel breathless. They cannot breathe. And very often they feel weakness."
The Treatment Bottleneck
Current treatment often involves catheter ablation, where a flexible tube is guided through blood vessels to the heart. Using heat or cold, doctors create tiny scars to block erratic electrical signals. However, capacity is severely strained. In Czechia alone, some 40,000 people are diagnosed annually, but hospitals can only treat about 10,000, leading to waiting lists of up to ten months. This shortfall is mirrored in health systems across the EU, driving the search for more efficient solutions.
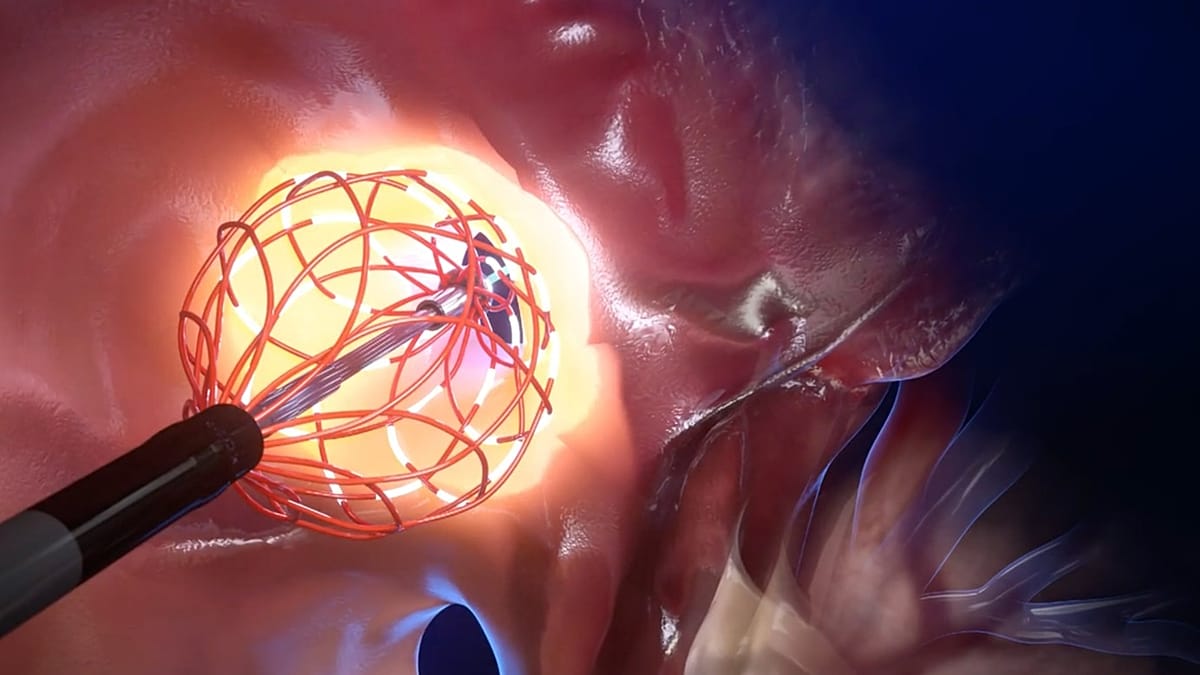
The new catheter, developed by the Czech firm BTL Industries, aims to break this bottleneck. Its key innovation is the application of microsecond energy pulses—lasting one-millionth of a second—targeted to destroy unhealthy heart tissue while sparing surrounding areas. "The procedure is safer because we apply microsecond pulses, which ideally destroy the myocardium and spare the other tissue, so the heart heals faster and better," says Martin Hanuliak, BTL's Head of Product Management.
This approach allows a physician to treat a larger area with a single, precise application. "You spend less time with the catheter. You have a single shot," notes Dr. Neužil. The company claims the technology could reduce ablation procedure time from several hours to as little as fifteen minutes, promising faster patient recovery, lower operational costs, and the ability to clear lengthy waiting lists.
Engineering Complexity and European Manufacturing
The device's sophistication lies in its miniaturised, moving components. "The main difference... is that ours is much more complex," explains product manager Jiří Dašek. "It contains very small parts that are less than one millimetre in size. The second difference is that our catheter moves, because in the past, the only catheters in use were straight and fixed."
BTL, a family-owned company that has grown into a global healthcare exporter, underscores the EU's industrial capacity in high-tech medtech. It employs 650 engineers among its 4,500 global staff. The firm's manufacturing facilities produce one million electronic boards annually, which are assembled into roughly 40,000 different medical devices. Upon regulatory approval, the new catheter and its control unit are planned for full EU-based production.
"Our turnover usually grows by 10% or 15% on average, depending on the year," says Chief Technology Officer Tomas Drbal. "If the company would like to keep growing, it needs to innovate." This catheter is at the forefront of that innovation drive. The ongoing clinical studies are a critical step toward the anticipated availability of the device on the European market by the beginning of 2028.
The development arrives amid broader European discussions on health security and strategic autonomy in critical sectors. While the continent focuses on external challenges, such as when the EU Energy Chief Warns of Prolonged Price Hikes from Middle East Conflict, internal innovation in healthcare technology remains a vital pillar of resilience. Similarly, as seen in the Greek Parliament Strips Immunity from 13 MPs in EU Farm Subsidy Fraud Investigation, robust systems and advanced tools are essential for addressing systemic pressures within the Union.
For millions of Europeans awaiting treatment for arrhythmia, the success of this Czech-led project could mean more than just a shorter procedure. It represents a tangible improvement in quality of life and a testament to the potential of European research, engineering, and manufacturing to solve pressing continental health challenges.